Research - Journal of Drug and Alcohol Research ( 2023) Volume 12, Issue 11
Pathogenesis of Bullous Pemphigoid
Cut Putri Hazlianda* and Widya Gabriella ManurungCut Putri Hazlianda, Departement of Dermatology Venereology and Aestethic, Universitas Sumatera Utara, Indonesia, Email: cut.putri@usu.ac.id
Received: 29-Dec-2023, Manuscript No. JDAR-23-123255; Editor assigned: 01-Jan-2024, Pre QC No. JDAR-23-123255 (PQ); Reviewed: 15-Jan-2024, QC No. JDAR-23-123255; Revised: 20-Jan-2024, Manuscript No. JDAR-23-123255 (R); Published: 27-Jan-2024, DOI: 10.4303/JDAR/236275
Abstract
The most prevalent autoimmune blistering illness of the mucous membranes and skin is Bullous Pemphigoid (BP). BP usually has the general appearance of tense skin blisters and pruritus. The disease is mediated by autoantibodies to the hemi-desmosomal proteins BP180 and BP230, which lead to blister formation. Inflammatory cells including eosinophils, monocytes, lymphocytes, neutrophils, and macrophages are present in the bullous cavities and upper dermis. Several proteinases, including Matrix Metalloproteinase (MMP)9, collagenase, plasmin, neutrophil elastase, are found in BP blister fluid, which have an important duty in the formation of sub-epidermal blister by degrading Extracellular Matrix (ECM) proteins. Eosinophils, Substance P (SP) and their receptors (NK1R, OSMRβ, and IL-31RA), IL-13, basophils, and periostin have the potential to cause itching in BP.
Keywords
Autoantibody; Bullous pemphigoid; Proteinase; Pruritus; Skin blister
Introduction
The most prevalent autoimmune bullous skin and mucous membrane disorder is Bullous Pemphigoid (BP). Normal signs of BP include pruritus and tight skin blisters. BP mostly targets the elderly [1].
The annual prevalence of BP ranges from 2.4 cases to 21.7 cases per million persons. BP often affects people over the age of 70 years to 90 years, but can also afflict young adults and children in drug-induced bullous pemphigoid. The population is at a high risk of dying from BP, with 1-year mortality rates ranging from 13% to 38%. Under the age of 75, women have a higher incidence than men, but after that point, men have a higher incidence. Infectious complications, the primary cause of death in people with BP, increase mortality rates [1,2].
Tight blisters, erythema, or urticarial plaques with intense itching are the clinical signs of BP. Non-bullous Pemphigoid (NBP), which is characterized by acute itching and crimson skin lesions, affects around one in every 5 individuals. Immune responses against 2 structural proteins in hemidesmosomes which are crucial for preserving skin integrity are the basis for BP and NBP. BP is a member of a diverse group of autoimmune subepithelial bullous disorders [2-4].
A histopathological examination of the perilesional skin of the most recent blister can be used to identify BP. The findings include dermal inflammatory infiltrates together with sub-epidermal blisters containing fibrin tissue and varied amounts of eosinophils and/or neutrophils [4]. Additionally, a direct immunofluorescence examination can spot IgG and/or C3 deposits that are smooth, linear, and continuous at the dermal-epidermal junction. Indirect Immunofluorescence (IIF) can demonstrate circulating IgG antibodies binding to the basal membrane [5].
Severe itching is one of the signs of BP, which lowers the patient’s quality of life. There are numerous itch mediators involved in itching, including cytokines/chemokines like Th2-associatedcytokines like interleukins (IL-31, IL-13, IL- 4), Thymic Stromal Lymphopoietin (TSLP), proteases and their associated receptors (Protease Activated Receptor-2 (PAR-2)), neuropeptides and receptors (Neurokinin-1 Receptor (NK1R) and Substance P (SP)), ion channels (vaniloid-1 (TRPV1) and Transient Receptor Potential Ankyin-1 (TRPA1)), and immune cells (mast cells, basophils, and eosinophils). Recently, it was discovered that eosinophils infiltrating BP lesions were expressing IL-31. However, the source of itching in BP is still mainly unknown. [3-5]. IgGclass autoantibodies against the antigens BP180 and BP230, 2 structural elements of the hemi-desmosome, induce blisters [4,5]. IgE autoantibodies can be found using immunofluorescence tests and Enzyme- Linked Immunoassay (ELISA) analyses and are also involved in the pathophysiology of illness. It is necessary to describe numerous more BP etiologyprocesses [2,4,5].
Discussion
Pathogenesis of bullous pemphigoid
Autoantibodies are known to bind to hemi-desmosomal antigens during the development of BP, which causes blisters formation. Studies using animal models show how BP autoantibodies develop [3,4].
Most of the precipitating factors of BP are unclear and occur sporadically. There are reportsthat BP can be triggered by Ultraviolet (UV) light, either UVB (or after psoralen therapy), ultraviolet A light, and radiation therapy. Additionally, some drugs like penicillamine, efalizumab, etanercept, and furosemide are linked to the development of BP. Recent research has demonstrated that immunological check point blocking with anti-programmed cell death receptor1 (anti-PD-1) therapy can also cause BP [5].
Bullous pemphigoid antigen
Direct Immunofluorescence (IF) demonstrates that patients with BP develop the tissue-bound autoantibodies against antigens from the skin’s Basement Membrane Zone (BMZ). Studies using microscopy immuno-electron pinpoint the BP antigen’s location on the hemi-desmosome. These autoantibodies attach to the hemi-desmosome’s extracellular surface and intracellular plaque. Two distinct antigens with molecular weights of 230 kDa and 180 kDa are present in BP [4,5]. The 230 kDa molecule is referred to PBAG1, BP230, or PBAG1e (indicating epidermal expression). BP230, a part of the protein plakin family, and is found in the hemi-desmosome’s intracellular plaque, in between keratin filaments. Keratin is held or held in place by BP230 on the hemi-desmosome. Animal models with BP230 deficiency show fragility in basal cells caused by breakdown of the keratin filament network [2,5,7]. While 180-kDa autoantigen is referred to type XVII collagen, PBAG2, or BP180. A transmembrane collagen protein called BP180 has an external carboxy terminal domain and an intracellular amino terminal domain. The extracellular domain is joined to the intracellular domain by anchoring filaments, which are both found in the hemi-desmosome plaque [5,7].
Complement cascade
The complement cascade’s function and alternate pathways in blister development are well recognized [2,7]. Complement deposits were visible in 83.1% of skin biopsies and supported by observational study in a large cohort of PB patients. The research also shown a connection between complement deposition, serological, and clinical disease activity [2].
Afterwards, Kasprick et al. [2,5,7], recently assessed the affects of monoclonal IgG2 antibody’s ability to prevent the activation of complement component 1s. The investigation confirmed that this antibody can inhibit the activation of the conventional complement system. Whereas, C5a Receptor 2 (C5aR2) appears to be protective, activated 5th complement Component in conjunction with receptor 1 is involved at the early stages of the disease. When the inflammatory process is fully developed, Reactive Oxygen Species (ROS) and proteases are produced.
Pathogenesis of sub-epidermal blister
The presence of circulating and binding autoantibodies against BP180 and BP230 is a distinguishing hallmark of bullous pemphigoid. The upper dermis and bullous cavities contain inflammatory cells such as eosinophils (the major cell type), neutrophils, lymphocytes, monocytes, and macrophages. Eosinophils, neutrophils, and Mast Cells (MC) can be present in the dermis in either an intact or degranulated state. Multiple inflammatory mediators found in the lesional or blister fluid can locally activate these cells. By destroying Extracellular Matrix (ECM) proteins, proteinases such plasmin, collagenase, neutrophil elastase, and Matrix Metalloproteinase (MMP)-9 play an important role in sub-epidermal blisters formation [4,5].
Data from both in vitro and in vivo experiments suggest that autoantibodies, notably those directed against BP180, start a whole chain of inflammatory events that result in tissue damage and blister formation. In order to destroy the physiological inhibitor of neutrophil elastase known as 1-proteinase inhibitor, plasmin activates the MMP-9 proenzyme, MMP-9, and then both enzymes. The dermalepidermal junction separates as a result of uncontrolled neutrophil elastase’s degradation of BP180 and other ECM constituents. A humanized BP180 mouse strain was created in order to immediately assess the toxicity of anti-BP180 IgG autoantibodies from BP patients. The murine BP180 domain or the equivalent domain is superseded by the human BP180or NC16A domain. IgG from BP patients causes sub-epidermal blisters to form [4,5] (Figure 1).
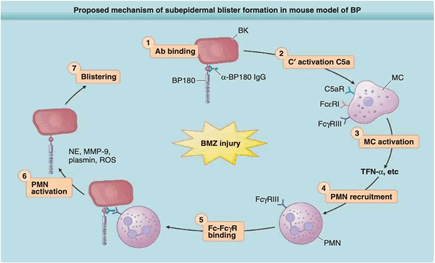
Figure 1: Proposed mechanism of sub-epidermal blister in BP
Recent studies have shown that deletion of the dominant BP180 epitope domain results in the development of anti- BP180 antibodies, which cause blistering and itching. The complicity of anti-BP230 autoantibodies in pemphigoid blisters has also been involved in several studies. Bullous pemphigoid is an antibody mediated autoimmune disease that depends on T cells in which autoreactive T lymphocytes can recognize BP180 coexisting with autoreactive B lymphocytes [5].
A number of additional sub-epidermal blister conditions, such as gestational pemphigoid, mucous membrane pemphigoid, linear IgA bullous dermatosis, and pemphigoid lichen planus, also have an autoimmune reaction to BP180. It’s probable that these conditions have immunopathological processes in common with BP [4].
Pathophysiology of itching in bullous pemphigoid
Eosinophils, Substance P (SP) and their receptors (NK1R, IL-31RA, and OSMRβ), IL-13, periostin, and basophils have the potential to cause itching in BP [3]. Multiple cell types, including sensory neurons and immunological cells (macrophages, T cells, and eosinophils), express SP. Sensory nerve fibers, keratinocytes, mast cells, and eosinophils all express NK1R. SP activates NK1Rexpressing cells, resulting in the generation of additional itch mediators and the sensation of itching. In their investigation, Hashimoto et al. found a correlation between the quantity of NK1R (+) and SP (+) cells and the level of itching in PB patients. The production of itch mediatorsby activated eosinophils might result in itching. The eosinophil-SP-NK1R axis has a crucial role in itch caused by BP. The itch-related cytokine IL-31, which is connected to the existence of anti-BP180 IgE antibodies, can also be secreted by eosinophils [3,5].
Non-bullous pemphigoid
Non Bullous Pemphigoid (NBP) is a subset of BP that lacks blisters but exhibits immunopathological symptoms similar to BP. Patients with NBP are frequently misdiagnosed as having dermatitis or a medication reaction. On primary skin that is not infected, erythematous lesions, papules, eczematous lesions, urticarial plaques, and pruritus are the most typical clinical signs [4].
IgG autoantibodies that target the hemi-desmosomal proteins BP180 and BP230 are created as part of the pathophysiology of NBP, which is strikingly similar to that of BP. The exact reason why blisters don’t form on NBP patients is unknown. It is believed that in NBP, BP230-predominant reactivity may be responsible for a less widespread disease response. In addition, complement activation was observed less frequently in NBP skin than in BP [4,5].
Bullous pemphigoid with other bullous diseases
Pemphigoid gestationis: Pemphigoid Gestationis (PG) is a form of pemphigoid that occurs in the second or 3rd trimester of pregnancy. Because PG’s antigenic targets resemble those of BP, it might be viewed as a pregnancyspecific variation of BP. According to one study, PG is brought on by the allogeneic gamma feto-placental unit in the mother’s placenta losing its protective function. MHCII antigen is not expressed on the trophoblast during a normal pregnancy. MHC II molecules, however, are aberrantly expressed in the placenta in PG patients. As a result, BP180 in the placenta is presented to the mother’s immune system.
Mucous membrane pemphigoid: In contrast to BP, mucosal areas are more likely to experience Mucous Membrane Pemphigoid (MMP), a chronic autoimmune sub-epidermal blister dermatosis with specific antigenic targets. The bullous lesions of MMP are produced by antibodies in the basement membrane zone. Based on the antigenic target involved, MMP has been broadly divided into 3 types:
1. Classic MMP-antibody targeting the C terminus domain of BP180;
2. Ocular MMP–antibody targeting the β4 subunit of the α6β4 integrin involved in the hemi-desmosome complex;
3. Anti-laminin 332 MMP-antibody targets laminin 332, a glycoprotein involved in the cohesion of the basement membrane zone.
Conclusion
Bullous pemphigoid is the most prevalent autoimmune blistering disease in adults. It usually presents as itchy, tense blisters, with an underlying urticarial plaque. The disease is mediated by autoantibodies to the hemidesmosomal proteins BP180 and BP230, which trigger an inflammatory cascade and lead to blister formation. The cells of inflammatory are present in the upper dermis and bullous cavities, including eosinophils (the predominant cell type), monocytes, lymphocyes, neutrophils, and macrophages. These cells activation can occur through the present of inflammatory mediators in the lesional or blister fluid. Some proteinases are found in BP blister fluid, such as Matrix Metalloproteinase (MMP)9, collagenase, plasmin, neutrophil elastase, which have an important role in subepidermal blister formation by degrading Extracellular Matrix (ECM) proteins. Eosinophils, Substance P (SP) and their receptors (NK1R, OSMRβ, and IL-31RA), IL- 13, basophils, and periostin have the potential to cause itching in BP.
Acknowledgement
None.
Conflict Of Interest
Authors have no conflict of interest to declare.
References
- K. Amber, D. Murrell, E. Schmidt, P. Joly, L. Borradori, Autoimmune subepidermal bullous diseases of the skin and mucosae: Clinical features, diagnosis, and management, Clin Rev Allergy Immunol, 54(2018):26-51.
- G. Genovese, G. Zenzo, E. Cozzani, E. Berti, M. Cugno, et al. New insights into the pathogenesis of bullous pemphigoid: 2019 update, Front Immunol, 10(2019):1-8.
- T. Hashimoto, C. Kursewicz, R. Fayne, S. Nanda, S. Shah, et al. Pathophysiologic mechanisms of itch in bullous pemphigoid, J Am Acad Dermatol, 83(2020):53-62.
- J. Meijer, A. Lamberts, J. Terra, Pemphigoid diseases affecting the skin, Springer, (2022):112-113.
- L. Goldsmith, S. Katz, B. Gilchrest, A. Paller, D. Leffell, et al. Fitzpatrick's dermatology in general medicine, 9th ed, McGraw Hill, New York, 2019.
- V. Di Lernia, D. Casanova, M. Goldust, C. Ricci, Pemphigus vulgaris and bullous pemphigoid: Update on diagnosis and treatment, Dermatol Pract Concept, 10(2020):50.
- J. Giang, M. Seelen, M. Van Doorn, R. Rissmann, E. Prens, et al. Complement activation in inflammatory skin diseases, Front Immunol, 16(2018).
Copyright: © 2023 Cut Putri Hazlianda, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

