Research Article: Journal of Drug and Alcohol Research (2022) Volume 11, Issue 4
Quercetin Compound Analysis to Develop Treatment for Dementia Associated with Alzheimerâs disease in Humans: In-silico Study
Amit Joshi1, Neha Vats2, Himanshu Singh1 and Vikas Kaushik1*2Deparment of Education, Lovely Professional University, India
Vikas Kaushik, Department of Bioengineering and Biosciences, Lovely Professional University, India, Email: vikas.14664@lpu.co.in
Received: 29-Mar-2022, Manuscript No. jdar-21-47627; Editor assigned: 31-Mar-2022, Pre QC No. jdar-21-47627 (PQ); Reviewed: 14-Apr-2022, QC No. jdar-21-47627; Revised: 19-Apr-2022, Manuscript No. jdar-21-47627 (R); Published: 26-Apr-2022, DOI: 10.4303/jdar/236174
Abstract
Background: Alzheimer’s disease is a memory loss problem associated mostly with elderly people, the cause of this disease is generally accumulation of beta-amyloid plaques around neuronal parts especially synaptic knobs, and also impaired synthesis of Tau proteins (Taupathy), that are mostly involved in holding micro-tubular assembly required for trafficking neurotransmitter containing vesicles.
New method: In this work, we perform computational analysis on the Quercetin molecule, an essential flavonoid that may stops the reaction participation tendency of beta-secretase biocatalyst, which could assist in the production of new treatments. Modern computational biology tools were deployed i.e., Swiss ADME for drug likeness, Auto-Dock Vina for molecular docking and Gromacs for molecular dynamics and simulation.
Results: Quercetin has good drug likeness as it follows Lipinski rule of five, also it perfectly docks with beta-secretase enzyme and shows binding energy of -9.8 Kcal/mol. For dry lab verifications, the docked complex of Quercetin and beta-secretase was submitted to molecular mechanics and simulation analyses.
Comparison with existing methods: No such methods were earlier used in this we designed computational approach to predict therapeutic nature of Quercetin compound for treatment of Alzheimer’s disease.
Conclusion: Our study reveals Quercetin molecule and beta-secretase enzyme have perfect binding, resulting in an inhibitory impact on beta- secretase enzyme activity. Our findings conclude that, Quercetin is beneficial and has anti-disease Alzheimer’s qualities.
Keywords
Alzheimer’s disease; Quercetin; Molecular dynamics simulation, Amyloid plaques; Taupathy
Introduction
In humans, Alzheimer’s disease is a neurological illness that impairs memory, thinking, moods, and mobility [1]. Alois Alzheimer, a German physician and pathologist, was the first to identify Alzheimer’s disease in 1906 [2]. The most obvious early indicator is difficulty recalling current events. As the illness advances, verbal abnormalities, disorientation, mood changes, lack of interest, self-neglect,and behavioural challenges may develop as possible signs [1]. It is frequently linked to ageing as a result of gerontology based physiological effects; the causes might include poor blood flow to the brain and impaired glucose metabolism. The illness is thought to be caused by plaques and tangles. Increased quantities of amyloid beta, which forms amyloid plaques outside the cell, and tau protein, that form neurofibrillary tangles inside the cell, are expected to exert Alzheimer’s disease by altering neuronal excitability and connection, leading in progressive loss of brain function [3]. Mutations in one of three genes that produce amyloid precursor protein (APP) and presenilins PSEN1 and PSEN2 cause rapidly progressive genetic Alzheimer’s disease [4]. The bulk of APP and presenilin gene mutations enhance the development of amyloid beta (A42), a small protein that is the main component of amyloid plaques [5]. Some mutations only alter the proportion of Amyloid42 to the other main kinds, especially Amyloid40, despite raising Amyloid42 concentrations [6]. Two further genes linked to inherited genetic Alzheimer’s disease were ABCA7 and SORL1 [7]. The most well-known genetic potential risk is the transfer of the apolipoprotein-E (APOE) gene [8]. At least one APOE4 allele is found in 40 to 80 percent of individuals affected with Alzheimer. In different allelic pair species, theAPOE4 gene carries the potential of illness by factor of three, while in similar allelic pair species, the probability is increased by factor of15 [9]. Newer genome wide interaction studies have discovered 19 gene regions that tend to control its harmful effect; CASS4, NME8, PTK2B, MS4A, and CD2AP are some of the alleles involved in this [10]. TREM2 genetic mutations have indeed been linked to a 3 to 5 fold elevated danger with Alzheimer [11]. One possibility reveals that TREM 2 variants CNS lose their capacity to regulate the amount of amyloid beta presence. A 2018 study divided Alzheimer’s disease into six domains, includes cognition, voice, spatial visual cognition, and mobility abilities, and added 30 single nucleotide polymorphisms (SNPs) to the list [12]. Many prior researches have shown that Quercetin has a significant function in the treatment of neuropathologies as well as a neuroprotective effect, making it useful in the treatment of neurodegenerative diseases [13].
Periplasmic amyloid beta deposition is the chief reason of Alzheimer’s disease, according to the amyloidosis concept, which was proposed in 1991 [14]. The fact, the persons with chromosomal triplication as in Down’s syndrome and associates additional genetic copy virtually always display early stages in Alzheimer just before 40 year age, and also the positioning in amyloid precursor protein loci at 21st Chromosomal set, support this notion [15]. In addition, APOE4, a particular isoform of apolipoprotein, is a significant genetic risk factor for Alzheimer’s disease. Although apolipoproteins help in the degradation of beta amyloid, certain isoforms (such as APOE4) are inactive, resulting in excessive amyloid accumulation in the brain [16]. Alzheimer’s disease is a protein misfolding illness, also known as a proteopathy that is characterized by plaque formation in the brain caused by incorrectly folded amyloid beta and tau proteins. Small peptides termed as amyloid betas, which are 39–43 amino acids long, make up plaques. Amyloid is a smaller portion of the bigger APP, an intermembrane glycoprotein that may cross a neuron’s membrane. It is required for neuron growth, survivorship, as well as in postinjury healing process [17]. Beta-Secretase collaborate mechanism that breaks down APP into smaller fragments in Alzheimer’s disease. Each of these fragments creates amyloid beta fibrils, which clump together and as a result inflammatory lesions become dense structures that grow outside of axons. Because of pathological tau protein accumulation (tauopathy) is common in Alzheimer Patient’s [18]. Neuronal cell does indeed have a cytoskeleton that is a microtubule based intrinsic support network serve as pathways for nutrients and chemicals to travel from the cell body to the axon ends. Tau is classified as a microtubule associated protein because it stabilizes microtubules when phosphorylated [19]. Tau reactive intermediates alterations in Alzheimer’s disease, becoming hyperphosphorylated, producing neurofibrillary tangles, and dissolving axonal transport by transposable factor dysregulation [20]. There are now five medications available to treat Alzheimer memory problems: 4acetylcholinesterase inhibitors (Tacrine-Galantamine and Donepezil), as well as1NMDA receptor antagonist Memantine [21]. EGb 761, a Ginkgo biloba extract, is commonly deployed for treatment of Alzheimer and other neuropsychiatric conditions [22]. EGb761 is an absorber of reactive oxygen species that enhances mitochondrial activity with serotonin and dopamine levels, making it neuro defensive. It has been shown in multiple studies that substantially enhance memory performance, tasks of daily life, and neuropsychiatric symptoms in persons with mild to severe dementia [23].
Quercetin (Q) is a flavonol found in plants that belongs to the flavonoid family of polyphenols. It may be found in a wide range of fruits, berries, leaves, nuts and grains; red onions and kale are the two prime examples of foods having high quercetin. The name comes from the Latin word quercetum, which means “oak woodland,” and refers to the oak species Quercus, and has been in use since 1857. It’s a polar auxin transport inhibitor that’s found in nature [24]. In crops, the main phenylpropanoid pathway transforms phenylalanine to 4-coumaroyl-CoA via phenylalanine ammonolyses, cinnamate4hydroxylase, and 4coumaroyl- CoAligase [25]. Part of 4coumaroylCoA is made up of 3 molecules of malonyl-CoA via 7,2′dihydroxy4′methoxyisoflavanolsynthase and create tetra hydroxychalcone. Tetrahydroxy chalcone is transformed into naringenin using chalcone isomerase. The enzyme flavonoid 3′-hydroxylase is responsible for converting naringenin to eriodictyol. Eriodictyol is transformed to dihydroquercetin via flavanone3hydroxylase, subsequently turned to quercetin via flavonol synthase [25]. In vitro, quercetin has indeed been proven to prevent other compounds from degrading, making it an antioxidant. It possesses a polyphenolic molecular composition that prevents degradation in vitro by acting as a scavenger of oxidative stress. Quercetin was already demonstrated to inhibit the PI3K/AKT cascade, resulting in the downregulation of the anti-apoptotic gene Bcl-w [26]. Quercetin containing plants is listed in (Table 1). Quercetin’s use in this study holds important role in Alzheimer’s control, Quercetin can protect neurons by inhibiting AChE enzymes, which increases acetylcholine levels and reduces Tau phosphorylation and amyloidbeta protein aggregation in Alzheimer. Quercetin is a potent inhibitor of JNK, PI- 3KAkt channels, acetylcholinesterase, nuclear translocation Factor, betasecretase-1/BACE-1 enzyme activity, and hyperphosphorylation. It also initiates production of AMP activated protein kinase that boosts the antioxidant key enzymes like superoxide dismutase-2 (AMPK). By reducing NADPHoxidase activity as well enhancing antioxidant activity for the enzymes such as superoxidedismutase2, this lowers the ros generation (ROS) [27].
Table1: Natural Sources of phytochemical Quercetin.
| Plant name | Family | Parts Of Plant | Content (mg/100gm) | References |
|---|---|---|---|---|
| Ruta graveolens | Rutaceae | Leaves | 45 | [27] |
| Allium cepa | Amaryllidaceae | Fruits | 32 | [28] |
| Mangifera indica | Anacardiaceae | Fruits | 22 | [29] |
| Solidagocanadensis | Asteraceae | Flowering parts | 23 | [30] |
| Phaleria macrocarpa | Thymelaceae | Seeds | 12 | [31] |
| Ginkgo biloba | Ginkgoaceae | Leaves | 68 | [32] |
| Crocus sativus | Iridaceae | Leaves | 81 | [33] |
In Figure 1 all the neuronal protective effects of Quercetin were clearly represented. In recent studies it was found that In-silico biology got advanced and assisted in predicting various phytochemical molecules against SARS-CoV2, like beta-sesquiphellandrene chemical derived from ginger binds to Spike protein and inhibits its interaction with ACE2 receptor of endothelial cells [28-34].
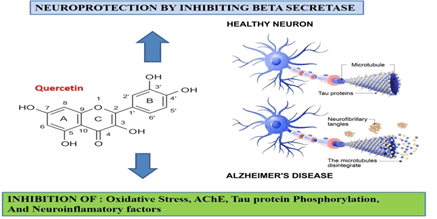
Figure 1: Effect of Quercetin on neuronal protection: Generalized role and effective role of Quercetin in neuroprotection.
Methodology
Quercetin ADME analysis
Quercetin properties were analyzed and its structure was retrieved from PubChem database, and swiss ADME web tool [35-37]. An ADME (adsorbent, delivery, metabolic and elimination) investigation for Quercetin being done by deploying swiss ADME server (http://www.swissadme.ch/). The Lipinski rule of five was satisfied for the validation [38], ADME analysis have also helped in predicting drug likeness. A feasible or oral route active drug must have no more 5 hydrogen bond donors (NH, OH bonds), no more than 10 hydrogen bond acceptors, a weight measurement of less than 500 Da, a log P less then equal 5 octanol water partition co-efficient, and no more than 10 rotational bonds, according to Lipinski rule.
Structure retrieval
The RCSB-PDB database (https://www.rcsb.org/search) was used to locate Beta secretase protein complexes. The drug’s architecture (quercetin) was received in sdf version from theNCBIPubChemrepository (https://pubchem. ncbi.nlm.nih.gov/) then afterwards it is changed to Pdb via OpenBabel [39].
Molecular docking
The association of protein and drug molecules was investigated using docking experiments. The PatchDock server was used to process the molecules, which were then sent to Fire Dock for screening. The PatchDock [40] tool can dock ligands with proteins for free in repositories that are open to the public access. The FireDock function on this server assists users in selecting the finest available complexes among thousands of docked complexes. It also provides a visual representation of the ligand’s interaction with the protein’s binding pocket, as well as an ACE (Atomic Contact Energy) of Docked_complexes. PyMOL [41], software was used to look at the H-bonds in between the ligand and receptor also shows the binding pocket; dependent on bond length and binding energy, this frequently validates the asassociation. Using the AutoDock Vina method for re-docking was done for measuring binding energies [42].
2D Visualization and H-bonding analysis
LigPlot+tool were deployed for analyzing 2D interaction pattern for drug and protein; it provides better pictorial representation of pharmacophore interacting with amino acid residues of considered enzyme [43].
Molecular dynamics simulations
GROMACS MD simulations were done deploying Linux based free Simulation software. An MD_Simulation for 100 ns was undertaken for the complex using Amber Force field in GROMACS. The docked groups were protonated and counterions were introduced accordingly overall charge set to 0 [44].
Results
Quercetin: Analysis of ADME properties
ADME analysis by deploying SwissADME tool shows Quercetin follows Lipinski rule of five, this clearly indicates its drug likeness. In Table 2 all ADME parameters are represented that were calculated by deriving chemical smile from PubChem database from NCBI (https://pubchem. ncbi.nlm.nih.gov/compound/Quercetin).
Table 2: SwissADME analysis report for Quercetin drug molecule.
| Sr.No. | ADME properties | Quercetin ADME outcomes |
|---|---|---|
| 1 | Molecular weight | 302.24g/mol |
| 2 | Chemical Formula | C15H10O7 |
| 3 | Number of hydrogen bond acceptors | 7 |
| 4 | Number of hydrogen bond donors | 5 |
| 5 | TPSA (Topological polar surface area) | 131.36Ǻ2 |
| 6 | Consensus log po/w (Partition coefficient between octanol and water) | 1.23 |
| 7 | Solubility | Soluble |
| 8 | Gastro-intestine absorbance | Yes |
| 9 | Blood Brain Barrier permeability | No |
| 10 | Cytochrome inhibitor | No |
| 11 | Lipinski Violations | Zero violations |
| 12 | Ghose Violations | Zero violations |
| 13 | Muegge violations | Zero violations |
| 14 | Drug likeness and Lead Likeness | Yes |
| 15 | Synthetic accessibility | 3.23 |
Structural retrieval of ligand and receptor
3D Structure of ligand i.e., drug molecule Quercetin downloaded from PubChem database in SDF format (PubChem Chemical ID for Quercetin: 5280343), then, using open babel tools, the drug structure file is translated to Pdb file. RCSB PDB database was used to download receptor (beta- secretase or BACE1) crystal structure, its PDB ID is 4LXM.
Molecular Interaction analysis
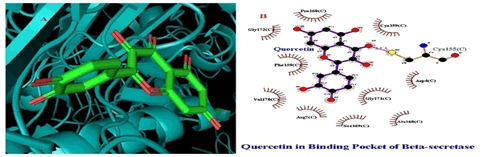
Molecular docking between ligand and receptor structural files was performed by using PatchDock and selection of best docked complexes was screened by applying Fire Dock screen. Fire Dock screens out top 10 best docked complexes out of 1000 docked complexes resulted after PatchDock analysis and top most docked complex was selected (Table 3). PatchDock provides ACE (Atomic Contact Energy) for interacting molecules while re-docking between ligand and receptor was performed by AutoDock vina tool to generate binding energy and also it provides finalized interaction patterns. In this docking analysis binding energy was determined to be -9.8 Kcal/mol between beta-secretase (Receptor) and Quercetin (Ligand). Interaction analysis was conducted by using LigPlot++ software, that generate better hydrogen bond pattern between Beta- secretase and Quercetin molecule (Figure 2).
Table 3: Atomic contact energy and global energy for FireDock screened docked complexes for top 10 results of Beta secretase and Quercetin compound molecular docking. (ACE=Atomic Contact Energy).
| Rank | Solution Number | Global Energy | Attractive VdW | Repulsive VdW | ACE (Kcal/mol) |
|---|---|---|---|---|---|
| 1 | 9 | -36.17 | -17.06 | 2.37 | -9.86 |
| 2 | 4 | -35.95 | -15.54 | 0.00 | -8.66 |
| 3 | 1 | -34.53 | -15.25 | 3.36 | -9.74 |
| 4 | 7 | -33.85 | -14.89 | 5.63 | -11.01 |
| 5 | 2 | -28.41 | -13.73 | 4.59 | -7.85 |
| 6 | 10 | -26.82 | -12.16 | 2.79 | -7.31 |
| 7 | 3 | -18.26 | -18.09 | 25.34 | -7.72 |
| 8 | 6 | -15.84 | -15.07 | 22.70 | -9.32 |
| 9 | 5 | -6.55 | -8.60 | 1.13 | 1.83 |
| 10 | 8 | 9.03 | -18.23 | 56.70 | -6.74 |
Figure 2: Molecular Interactions between Quercetin and Beta secretase: A. 3D interaction pattern visualized in PyMOL. 2D interaction revealing hydrogen bond (2.5Ǻ) between Cysteine amino acid residue at 155th position and Quercetin molecule by using LigPlot ++.
Molecular dynamics simulations
Then, using open babel tools, the drug structure file is translated to Pdb file. For both the docked and free beta- secretase models in Figure 3, trajectory Analysis shows (RMSD) and (RMSF) plots. The¬ RMSD values were found to be in the 0.2-0.6 nm range, and the RMSF values were found to be in the 0.1-0.5 nm range for docked complex of Quercetin and Beta-secretase. The MD analysis for 100 ns time scale clearly indicates more stability of considered docked complex.
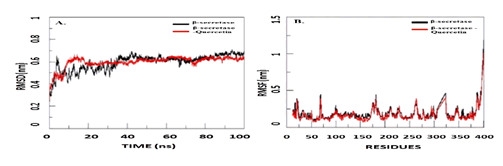
Figure 3: Molecular dynamic simulation results: A. RMSD and B. RMSF plots for docked (with Quercetin) and free beta-secretase.
Discussion
Quercetin a natural pharmaco active compound present in many plants was found to have neuroprotective role in many recent studies, our research not only provides dry lab investigations but also neuronal cell line testing of this therapeutic molecule [45,46]. In our study ADME analysis of Quercetin molecule revealed that it follows Lipinski rule of five and exhibit drug likeness, also molecular docking experiments revealed perfect binding of this therapeutic molecule with beta-secretase enzyme, which is responsible for production of beta-amyloid plaques at synaptic knobs [47]. Molecular dynamics and simulation studies reveals that Quercetin perfectly binds the binding pocket of beta-secretase enzyme hence brought inhibitory effect on its activity. Alzheimer’s disease and other neurodegenerative illnesses are caused by a range of pathologies and etiologies. According to an increasing amount of data, free radical induced toxicity, oxidative damage, mitochondrial_ malfunction, and proinflammatory cytokines can all play a role in the development of neurodegeneration [48]. Microglia, a kind of brain cell, has been implicated in neuropathological disorders. By suppressing microglial activation, vitamin-E and minocycline, on either hand, have been proven to have neuroprotective qualities [49]. These molecular processes through which Amyloid Precursor Protein with secretase form transitory or stable complexes are, on the other hand largely unexplored [50]. Quercetin dramatically reduced Amyloid Precursor Protein breakdown via secretase and Amyloid beta production in a recombinant SH-SY5Y control cells, according to many previous studies. The neuroprotective impacts of quercetin are mainly due to possible cytokine up and/ or down regulation via the nuclear factor (erythroidderived2) like 2(Nrf2), Paraoxonase2, cJun Nterminal kinase (JNK), ProteinkinaseC, Mitogen activated protein kinase (MAPK), and PI3K/Akt signaling pathways [51]. In many recent studies it was found that computational drug designing and vaccine development strategies were successfully conducted for therapeutic development against pathogens like Candida fungus, Dengue Virus, Nervous necrosis virus, HCMV virus, also Canine circo virus; and these In-silico studies were very time and money efficient that open new avenues for computer based drug designing [52-56].
Conclusion
In current research, Quercetin possible drug candidate tested successfully via computational biology experiments. Our study reveals that Quercetin molecule perfectly binds with beta-secretase enzyme and exhibited binding energy of -9.8 Kcal/mol, which is required for producing inhibitory effect on beta-secretase activity. This β-secretase enzyme has been characterized in order to help in the formulation of a new anti-Alzheimer therapy for the pharmaceutical or food supplement industries. Our research clearly indicates that Quercetin can effectively inhibit beta-secretase and stops the formation of amyloid plaques. Our study reveals that Quercetin use would be worthy and holds therapeutic properties against Alzheimer’s disease. This study also opens door for testing of Quercetin on model organisms for determining effective dosage patterns as a future scope of our research.
Author Statement
AJ, NV, and VK conducted the research, and wrote manuscript. All authors were equally involved in all aspects of manuscript preparation. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Ethical Statement
No experiments conducted on any living or dead animal in this research study.
Acknowledgement
We would like to acknowledge School of bioengineering and biosciences, Lovely professional University, Punjab (India) for providing facilities for conducting this research.
References
- Burns, Slliffe, Alzheimer’s disease,BMJ,338(2009), 158.
- N.C. Berchtold, C. W. Cotman, Evolution in the conceptualization of dementia and Alzheimer’s disease: Greco-Roman period to the 1960s,Neurobiol Aging,19(1998), 173-189.
[Crossref] [Google Scholar] [PubMed]
- Tackenberg, L.Kulic, R.M.Nitsch, Familial Alzheimer’s disease mutations at position 22 of the amyloid β-peptide sequence differentially affect synaptic loss, tau phosphorylation and neuronal cell death in an ex vivo system,PloS one,15(2020), e0239584.
[Crossref] [Google Scholar] [PubMed]
- S.C. Waring, R.N. Rosenberg, Genome-wide association studies in Alzheimer disease, Arch Neurol,65(2008), 329-334.
[Crossref] [Google Scholar] [PubMed]
- D.J. Selkoe,Translating cell biology into therapeutic advances in Alzheimer's disease,Nature,399(1999), A23-A31.
[Crossref] [Google Scholar] [PubMed]
- D.R. Borchelt, G. Thinakaran, C.B. Eckman, M.K. Lee, F. Davenport, et al.Familial Alzheimer's disease–linked presenilin 1 variants elevate Aβ1–42/1–40 ratio in vitro and in vivo,Neuron,17(1996), 1005-1013.
[Crossref] [Google Scholar] [PubMed]
- J.H. Kim,Genetics of Alzheimer's disease,Dement Neurocogn Disord,17(2018), 131.
[Crossref] [ResearchGate]
- W. J. Strittmatter, A. M. Saunders, D. Schmechel, M. Pericak-Vance, J. Enghild, Apolipoprotein E: High-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease,Proc Nat Acad Sci,90(1993), 1977-1981.
[Crossref] [Google Scholar] [PubMed]
- R.W. Mahley, K.H. Weisgraber, Y. Huang, Apolipoprotein E4: A causative factor and therapeutic target in neuropathology, including Alzheimer’s disease,Proc Nat Acad Sci,103(2006), 5644-5651.
[Crossref] [Google Scholar] [PubMed]
- J.C. Lambert, C. A. Ibrahim-Verbaas, D. Harold, A. C. Naj, R. Sims, et al. European Alzheimer’s Disease I, Genetic, Environmental Risk in Alzheimer’s D, Alzheimer’s Disease Genetic C, Cohorts for H, Aging Research in Genomic E. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease,Nat Genet,45(2013), 1452-1458.
[Google Scholar] [PubMed]
- T. Jonsson, H. Stefansson, S. Steinberg, I. Jonsdottir, P.V. Jonsson, et al. Variant of TREM2 associated with the risk of Alzheimer’s disease,New England J Med,368(2013), 107-116.
[Crossref] [Google Scholar] [PubMed]
- S. Mukherjee, J. Mez, E.H. Trittschuh, A.J. Saykin, L.E. Gibbons, et al. Genetic data and cognitively defined late-onset Alzheimer’s disease subgroups,Mol Psych,25(2020), 2942-2951.
[Crossref] [Google Scholar] [PubMed]
- Z. H. Zhang, L. C. V. Cheang, M. W. Wang, S.M.Y. Lee, Quercetin exerts a neuroprotective effect through inhibition of the iNOS/NO system and pro-inflammation gene expression in PC12 cells and in zebrafish,Int J Mol Med,27(2011), 195-203.
[Crossref] [Google Scholar] [PubMed]
- J. Hardy, D. Allsop, Amyloid deposition as the central event in the aetiology of Alzheimer's disease,Trends Pharmacol Sci,12(1991), 383-388.
[Crossref] [Google Scholar] [PubMed]
- M. Nistor, M. Don, M. Parekh, F. Sarsoza, M. Goodus, et al. Alpha-and beta-secretase activity as a function of age and beta-amyloid in Down syndrome and normal brain,Neurobiol Aging,28(2007), 1493-1506.
[Crossref] [Google Scholar] [PubMed]
- D. Games, D. Adams, R. Alessandrini, R. Barbour, P. Borthelette, et al. Alzheimer-type neuropathology in transgenic mice overexpressing V717F β-amyloid precursor protein,Nature,373(1995), 523-527.
[Crossref] [Google Scholar] [PubMed]
- C. Priller, T. Bauer, G. Mitteregger, B. Krebs, H. A. Kretzschmar, et al. Synapse formation and function is modulated by the amyloid precursor protein,J Neurosci,26(2006), 7212-7221.
[Crossref] [Google Scholar] [PubMed]
- N. M. Hooper, Roles of proteolysis and lipid rafts in the processing of the amyloid precursor protein and prion protein,Biochem Soc Transac,33(2005), 335-338.
[Crossref] [Google Scholar] [PubMed]
- S. Camero, M.J. Benítez, R. Cuadros, F. Hernández, J. Ávila, et al. Thermodynamics of the interaction between Alzheimer's disease related tau protein and DNA,PLoS One,9(2014), e104690.
[Crossref] [Google Scholar] [PubMed]
- W. Sun, H. Samimi, M. Gamez, H. Zare, B. Frost, Pathogenic tau-induced piRNA depletion promotes neuronal death through transposable element dysregulation in neurodegenerative tauopathies,Nature Neurosci,21(2018), 1038-1048.
- H.A. Fink, E.J. Linskens, R. MacDonald, P.C. Silverman, J.R. McCarten, et al. Benefits and harms of prescription drugs and supplements for treatment of clinical Alzheimer-type dementia: A systematic review and meta-analysis,Ann Int Med,172(2020), 656-668.
[Crossref] [Google Scholar] [PubMed]
- K. McKeage, K.A. Lyseng-Williamson, Ginkgo biloba extract egb 761® in the symptomatic treatment of mild-to-moderate dementia: A profile of its use,Drugs Ther Perspectives,34(2018), 358-366.
- N. Kandiah, P.A. Ong, T. Yuda, L. L. Ng, K. Mamun, et al. Treatment of dementia and mild cognitive impairment with or without cerebrovascular disease: Expert consensus on the use of Ginkgo biloba extract, EGb 761®,CNS Neurosci Ther,25(2019), 288-298.
[Crossref] [Google Scholar] [PubMed]
- C. Fischer, V. Speth, S. Fleig-Eberenz, G. Neuhaus, Induction of zygotic polyembryos in wheat: Influence of auxin polar transport,The Plant Cell,9(1997), 1767-1780.
[Crossref] [Google Scholar] [PubMed]
- B. Winkel-Shirley, Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology,Plant Physiol,126(2001), 485-493.
[Crossref] [Google Scholar] [PubMed]
- M.L. Hartman, M. Czyz, BCL-w: Apoptotic and non-apoptotic role in health and disease,Cell Death Disease,11(2020), 1-16.
[Crossref] [Google Scholar] [PubMed]
- H. Khan, H. Ullah, M. Aschner, W.S. Cheang, E.K. Akkol, Neuroprotective effects of quercetin in Alzheimer’s disease,Biomol,10(2020), 59.
[Crossref] [Google Scholar] [PubMed]
- M. Choudhary, V. Kumar, H. Malhotra, S. Singh, Medicinal plants with potential anti-arthritic activity,J Intercult Ethnopharmacol,4(2015), 147.
[Crossref] [Google Scholar] [PubMed]
- T.M. Henagan, W.T. Cefalu, D.M. Ribnicky, R.C. Noland, K. Dunville, et al. In vivo effects of dietary quercetin and quercetin-rich red onion extract on skeletal muscle mitochondria, metabolism, and insulin sensitivity,Genes Nutri,10(2015), 2.
[Crossref] [Google Scholar] [PubMed]
- M. Gondi, U. P. Rao, Ethanol extract of mango (Mangifera indica L.) peel inhibits α-amylase and α-glucosidase activities, and ameliorates diabetes related biochemical parameters in streptozotocin (STZ)-induced diabetic rats,J Food Sci Technol,52(2015), 7883-7893.
[Crossref] [Google Scholar] [PubMed]
- P. Apati, K. Szentmihályi, A. Balázs, D. Baumann, D., M. Hamburger, et al. HPLC analysis of the flavonoids in pharmaceutical preparations from Canadian goldenrod (Solidago canadensis),Chromatographia,56(2002), S65-S68.
- R. Altaf, M. Z. B. Asmawi, A. Dewa, A. Sadikun, M. I. Umar, Phytochemistry and medicinal properties of Phaleriamacrocarpa (Scheff.) Boerl. Extracts,Pharma Rev,7(2013), 73.
[Crossref] [Google Scholar] [PubMed]
- P.C. Chan, Q. Xia, P.P. Fu, Ginkgo biloba leave extract: Biological, medicinal, and toxicological effects,J Environ Sci Health,25(2007), 211-244.
[Crossref] [Google Scholar] [PubMed]
- J. Mottaghipisheh, M. M. Sourestani, T. Kiss, A. Horváth, B. Tóth, et al. Comprehensive chemotaxonomic analysis of saffron crocus tepal and stamen samples, as raw materials with potential antidepressant activity,J Pharma biomedical Anal,184(2020), 113183.
[Crossref] [Google Scholar] [PubMed]
- A. Joshi, G.S. Krishnan, V. Kaushik, Molecular docking and simulation investigation: Effect of beta-sesquiphellandrene with ionic integration on SARS-CoV2 and SFTS viruses,J Genetic Eng Biotechnol,18(2020), 1-8.
[Crossref] [Google Scholar] [PubMed]
- S. Kim, J. Chen, T. Cheng, A. Gindulyte, J. He, S. He, et al. PubChem 2019 update: Improved access to chemical data,Nucleic acids Res,47(2019), D1102-D1109.
- A. Daina, O. Michielin, V. Zoete, SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules,Sci Rep,7(2017), 1-13.
[Crossref] [Google Scholar] [PubMed]
- C.A. Lipinski, Lead-and drug-like compounds: The rule-of-five revolution,Drug Discovery Today: Technol,1(2004), 337-341.
[Crossref] [Google Scholar] [PubMed]
- N. M. O'Boyle, M. Banck, C. A. James, C. Morley, T. Vandermeersch, G.R. Hutchison, Open Babel: An open chemical toolbox,J Cheminfor,3(2011), 1-14.
[Crossref] [Google Scholar] [PubMed]
- D. Schneidman-Duhovny, Y. Inbar, R. Nussinov, H.J. Wolfson, PatchDock and SymmDock: Servers for rigid and symmetric docking,Nucleic acids Res,33(2005), W363-W367.
[Crossref] [Google Scholar] [PubMed]
- M.A. Lill, M. L. Danielson, Computer-aided drug design platform using PyMOL,J Comp-aided Mol Des,25(2011), 13-19.
[Crossref] [Google Scholar] [PubMed]
- O. Trott, A.J. Olson, AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading,J Comput Chem,31(2010), 455-461.
[Crossref] [Google Scholar] [PubMed]
- R.A. Laskowski, M.B. Swindells, LigPlot+: Multiple ligand–protein interaction diagrams for drug discovery, J Chem Inf Model,51(2011), 2778-2786.
[Crossref] [Google Scholar] [PubMed]
- D. Van Der Spoel, E. Lindahl, B. Hess, G. Groenhof, A.E. Mark, et al., GROMACS: Fast, flexible, and free,J Comput Chem,26(2005), 1701-1718.
[Crossref] [Google Scholar] [PubMed]
- E. Akyuz, Y.N. Paudel, A.K. Polat, H.E. Dundar, E. Angelopoulou, Enlightening the neuroprotective effect of quercetin in epilepsy: From mechanism to therapeutic opportunities,Epilepsy Behav,115(2021), 107701.
[Crossref] [Google Scholar] [PubMed]
- H. Shen, Y. Zheng, R. Chen, X. Huang, G. Shi, Neuroprotective effects of quercetin 3-O-sophoroside from Hibiscus rosa-sinensis Linn. on scopolamine-induced amnesia in mice,J Funct Foods,76(2021), 104291.
- A.B. Reiss, N. Montufar, J. DeLeon, A. Pinkhasov, I. H. Gomolin, A. D. Glass, et al. Alzheimer Disease Clinical Trials Targeting Amyloid: Lessons Learned From Success in Mice and Failure in Humans,Neurol,26(2021), 52-61.
[Crossref] [Google Scholar] [PubMed]
- P. J. Crouch, S.M.E. Harding, A.R. White, J. Camakaris, A.I. Bush, et al. Mechanisms of Aβ mediated neurodegeneration in Alzheimer's disease,Int J Biochem Cell Biol,40(2008), 181-198.
[Crossref] [Google Scholar] [PubMed]
- J.K. Ryu, S. Franciosi, P. Sattayaprasert, S.U. Kim, J.G. McLarnon, Minocycline inhibits neuronal death and glial activation induced by β-amyloid peptide in rat hippocampus,Glia,48(2004), 85-90.
[Crossref] [Google Scholar] [PubMed]
- A.K. Ghosh, M. Brindisi, J. Tang, Developing β-secretase inhibitors for treatment of Alzheimer’s disease,J Neurochem,120(2012), 71-83.
[Crossref] [Google Scholar] [PubMed]
- E. Zaplatic, M. Bule, S.Z.A. Shah, M.S. Uddin, K. Niaz, Molecular mechanisms underlying protective role of quercetin in attenuating Alzheimer's disease,Life Sci,224(2019), 109-119.
[Crossref] [Google Scholar] [PubMed]
- N. Akhtar, A. Joshi, V. Kaushik, M. Kumar, M.A.U. Mannan, In-silico design of a multivalent epitope-based vaccine against Candida auris,Microbial Pathogenesis,155(2021), 104879.
[Crossref] [Google Scholar] [PubMed]
- S. Krishnan, A. Joshi, V. Kaushik, T cell epitope designing for dengue peptide vaccine using docking and molecular simulation studies, Mol Simul,46(2020), 787-795.
- A. Joshi, D.C. Pathak, M.A.U. Mannan, V. Kaushik, In-silico designing of epitope-based vaccine against the seven banded grouper nervous necrosis virus affecting fish species,Net Modeling Analysis in Health Informatics and Bioinformatics,10(2021), 1-12.
[Crossref] [Google Scholar] [PubMed]
- N. Akhtar, A. Joshi, J. Singh, V. Kaushik, Design of a Novel and Potent Multivalent Epitope Based Human Cytomegalovirus Peptide Vaccine: An Immunoinformatics Approach, J Mol Liquids, 116586(2021).
- P. Jain, A. Joshi, N. Akhtar, S. Krishnan, V. Kaushik, An immunoinformatics study: Designing multivalent T-cell epitope vaccine against canine circovirus,J Genetic Eng Biotechnol,19(2021), 1-11.
[Crossref] [Google Scholar] [PubMed]
Copyright: © 2022 Vikas Kaushik, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.